
|
| About the Journal |
| Aims and Scope |
| Journal Information |
| Editorial Board |
| Best Practice |
| Subscriptions |
| Contact Us |


|
| About the Journal |
| Aims and Scope |
| Journal Information |
| Editorial Board |
| Best Practice |
| Subscriptions |
| Contact Us |

This article has been corrected. See "Correction of dosage of the drug: Post-infectious basal ganglia encephalitis and axonal variant of Guillain-Barré syndrome after COVID-19 infection: an atypical case report" in Volume 25 on page 54. AbstractNeurological complications attributed to coronavirus disease-19 (COVID-19) infection have been reported including acute disseminated encephalomyelitis, Guillain-Barré syndrome, and so on. Herein, we report a 49-year-old woman presented with acute encephalopathy and paraplegia simultaneously after COVID-19 infection. Brain magnetic resonance imaging (MRI) showed symmetric hyperintense basal ganglia lesions on T2-weighted imaging. Cerebrospinal fluid pleocytosis, motor axonal neuropathy and enhancement of conus medullaris nerve roots on spine MRI were observed. We treated her with high-dose corticosteroid and intravenous immunoglobulin.
Encephalitis/encephalopathy and Guillain-Barré syndrome (GBS) are known to be the most prevalent, immune-mediated neurological complications of coronavirus disease 2019 (COVID-19) in the central nervous system (CNS) and the peripheral nervous system (PNS).1 A systematic review and meta-analysis found that the pooled GBS prevalence was 0.15% (95% confidence interval [CI], 0-0.49%) and that the average incidence of encephalitis was 0.215% (95% CI, 0.056-0.441%) in association with COVID-19.2,3 The present authors encountered a patient who presented with the involvement of both nervous systems simultaneously, with atypical brain magnetic resonance imaging (MRI) showing symmetrical T2-weighted imaging (T2WI) and fluid-attenuated inversion recovery (FLAIR) hyperintensities without contrast enhancement in the basal ganglia (BG).
CASEA 49-year-old female was admitted to our hospital due to drowsiness, inability to sit and walk, and paraplegia that appeared 15 days after SARS-CoV-2 infection. The patient was infected in March 2022 when the number of SARS-CoV-2 infections exploded in South Korea and associated symptoms were mild fever and myalgia. A neurological examination revealed that she was drowsy and disorientated for time, place, and person. A cranial nerve function test showed dysarthria, dysphagia, absent gag reflex, and gaze-evoked nystagmus. Muscle strength of the upper extremities was against gravity and there was some resistance bilaterally, but the lower extremities were completely paraplegic and flaccid. Myotatic reflexes of the upper extremities were normal, but they could not be elicited in the lower extremities. Sensory functions were not evaluated due to the patient’s somnolence and inattention. Although the patient was obtunded, her respiration was stable and she did not require tracheal intubation or ventilation.
Blood tests showed normal liver function, renal function, and electrolyte, ammonia, and glucose levels, except for mild leukocytosis (13,070/mm3) and elevated high-sensitivity C-reactive protein (5.91 mg/dL; reference range, 0.05-0.5 mg/dL). Brain MRI showed an edematous hyperintense lesion in the bilateral BG including the putamen, caudate and globus pallidus in T2WI and FLAIR imaging (Fig. 1A, B). Gadolinium contrast did not reveal any enhancement, and diffusion restriction was not observed (Fig. 1C). Electroencephalography (EEG) showed generalized slow waves. The following abnormal results were obtained in a cerebrospinal fluid (CSF) analysis: opening pressure of 200 mmHg, red blood cell count of 200/mm3, white blood cell count of 92/mm3 with lymphocyte predominance (98%), protein level of 84.1 mg/dL, glucose at 50 mg/dL, normal CSF immunoglobulin (Ig) G index, and positivity for CSF-restricted oligoclonal band. CSF pleocytosis with protein elevation was suggestive of CNS infection/inflammation. However, CSF culturing and polymerase chain reaction for viruses (herpes simplex virus, Epstein-Barr virus, varicella zoster virus, cytomegalovirus, enterovirus, and Japanese encephalitis virus), mycobacterium tuberculosis, bacteria, and fungus produced negative findings, ruling out infectious encephalitis. Unfortunately, the presence of SARS-CoV-2 could not be tested in CSF. Serological antibodies (Abs) against nuclear, thyroid peroxidase, thyroglobulin, aquaporin-4, and gangliosides (GQ1b, GM1, and GD1b), and paraneoplastic antineuronal Abs (Hu, Yo, Ri, amphiphysin, CV2, PNMA2 [Ma2/Ta], recoverin, SOX1, and Titin) were negative in the serum. Screening for autoimmune encephalitis with Abs against neuronal surface antigens (NMDAR, AMPAR, DPPX, LGI1, CASPR2, and GABABR) yielded negative results in both serum and CSF. In addition to negative paraneoplastic autoantibodies in the serum, computed tomography of the head, neck, chest, and abdomen did not reveal any remarkable abnormality related to malignancy. Spine MRI showed normal cord signal intensities with multiple dot-like enhancements along the conus medullaris (Fig. 2). Nerve conduction studies (NCSs) performed on days 7 and 21 after admission revealed axonal motor neuropathy (Table 1).
Based on the clinical, electrophysiological, laboratory, and imaging findings, the patient was diagnosed with postinfectious encephalitis and an axonal variant of GBS. In a differential diagnosis of MRI findings, toxic-metabolic encephalopathy and osmotic demyelination syndrome were excluded based on the laboratory results and clinical course. The patient was treated with intravenous ig (IVIg) (0.4 mg/kg/day for 5 consecutive days) and high-dose corticosteroid (1 g/day for 3 days followed by slowly tapering). This treatment and rehabilitation resulted in her consciousness, orientation, cranial nerve functions, and upper extremity functions returning to normal, but the paraplegia in her legs did not improve at the time of discharge. However, 6 months after discharge, her legs were strong enough to walk slowly with a walker and myotatic reflexes of the knees were observed. On Follow-up brain MRI, T2 hyperintensities in both BG were disappeared and mild brain atrophy was developed (Fig. 1D).
DISCUSSIONThere have been reports on individual neurological complications occurring in the CNS or PNS during or after COVID-19 infection due to direct viral invasion into the nervous system, neurological manifestation of COVID-19 as a systemic illness, and parainfectious/postinfectious inflammation mediated.1 However, to our knowledge, the present patient is the first post-COVID-19 case with neurological complications involving the CNS and PNS simultaneously, in the form of encephalitis and GBS. There are several interesting and controversial aspects related to the present diagnosis. Based on the altered mental status, awareness impairment, and the results of EEG and CSF analysis, the authors had no doubt that encephalitis was present.
The finding from brain MRI in our patient, which was mild edematous hyperintense BG lesions in T2WI and FLAIR imaging bilaterally, did not fit the acute disseminated encephalomyelitis (ADEM), viral encephalitis, autoimmune encephalitis, or neuroimaging frequently seen in COVID-19 patients. Brain MRI of viral and autoimmune encephalitis typically shows an area of T2WI and FLAIR hyperintensities involving both the cortex and the white matter with various degrees of contrast enhancement in the limbic lobe, temporal lobe, and brainstem.4 ADEM appears (by definition) as multiple bilateral but asymmetric T2WI/FLAIR hyperintensities involving the cortical junction between gray and white matter with a smudged margin and variable contrast enhancement.5 Neuroimaging findings in COVID-19 patients include a specific diagnosis (e.g., acute stroke or posterior reversible encephalopathy syndrome) or specific imaging features (diffuse leukoencephalopathy, leptomeningeal enhancement, cortical FLAIR signal abnormality, or microhemorrhages).6
Bilateral symmetric BG lesions that are hyperintense in T2WI/FLAIR imaging include genetic, acquired, metabolic, and toxic pathologies for the differential diagnosis, none of which corresponded to our case.7 Autoimmune BG encephalitis with neuroimaging results revealing bilateral lesions in the BG with or without contrast enhancement and diffusion restriction has also been reported recently, which are similar to our imaging results.8,9 This usually manifests as acute-onset parkinsonism, chorea, dystonia, and behavioral change, with very mild CSF pleocytosis. However, the absence of movement disorder, relatively high CSF white blood cell count, negative Abs serology for neuron and neuronal surface antigens, and the recent history of COVID-19 infection made it likely that our patient had postinfectious BG encephalitis rather than autoimmune BG encephalitis.
The patient’s clinical presentation showing complete paraplegia with absent myotatic jerks was consistent with a diagnostic certainty of level 2 according to the Brighton criteria for GBS, except for CSF pleocytosis, and nerve-root enhancement in the conus medullaris also supports the presence of GBS in our patient.10 There have been reports of an atypical type of GBS called the paraparetic variant, which is classified as a localized form of acute motor axonal neuropathy (AMAN).11 Moreover, a case of paraparetic GBS of AMAN with CSF pleocytosis exceeding 50/mm3 has been reported,12 like in our case. Transverse myelitis could be ruled out by the normal spinal cord signals, absent sensory symptoms, and negative pathological reflexes. Critical illness polyneuropathy seems less likely considering the acute symptom onset and the abnormal NCS findings being restricted to motor nerves of the lower extremities.
There is no treatment consensus for para-/postinfectious encephalitis after SARS-CoV-2 infection, although IVIg and corticosteroid can be administered due to the possibility of an immune-mediated mechanism like in GBS. The data on treatment outcomes of post-COVID-19-associated severe complications in the CNS and PNS are also sparse, with instead only individual case reports that have had various prognoses. It seems necessary to organize and analyze cases in detail about post-COVID-19-associated neurological complications in order to broaden the horizons of physicians on various aspects of complications including such an atypical manifestation like in the present case.
In summary, SARS-CoV-2 infection can produce simultaneous encephalitis and GBS probably via a postinfectious immune-mediated process and its associated neuroimaging abnormalities can appear in the BG that resemble the characteristics of autoimmune BG encephalitis.
Supplementary MaterialSupplementary Materials can be found with this article online https://doi.org/10.14253/acn.2022.24.2.101
Supplementary Table 1.Results of both consecutive sensory nerve conduction studies are normal
Fig. 1.Brain magnetic resonance imaging (MRI) showed abnormal hyperintense signals (yellow arrows) in the bilateral basal ganglia in axial T2-weighted imaging (T2WI) (A) and fluid-attenuated inversion recovery imaging (FLAIR) (B) with no gadolinium contrast enhancement in the corresponding areas (C), and these signals disappeared with instead mild brain atrophy developed on follow-up MRI after 6 months (D). 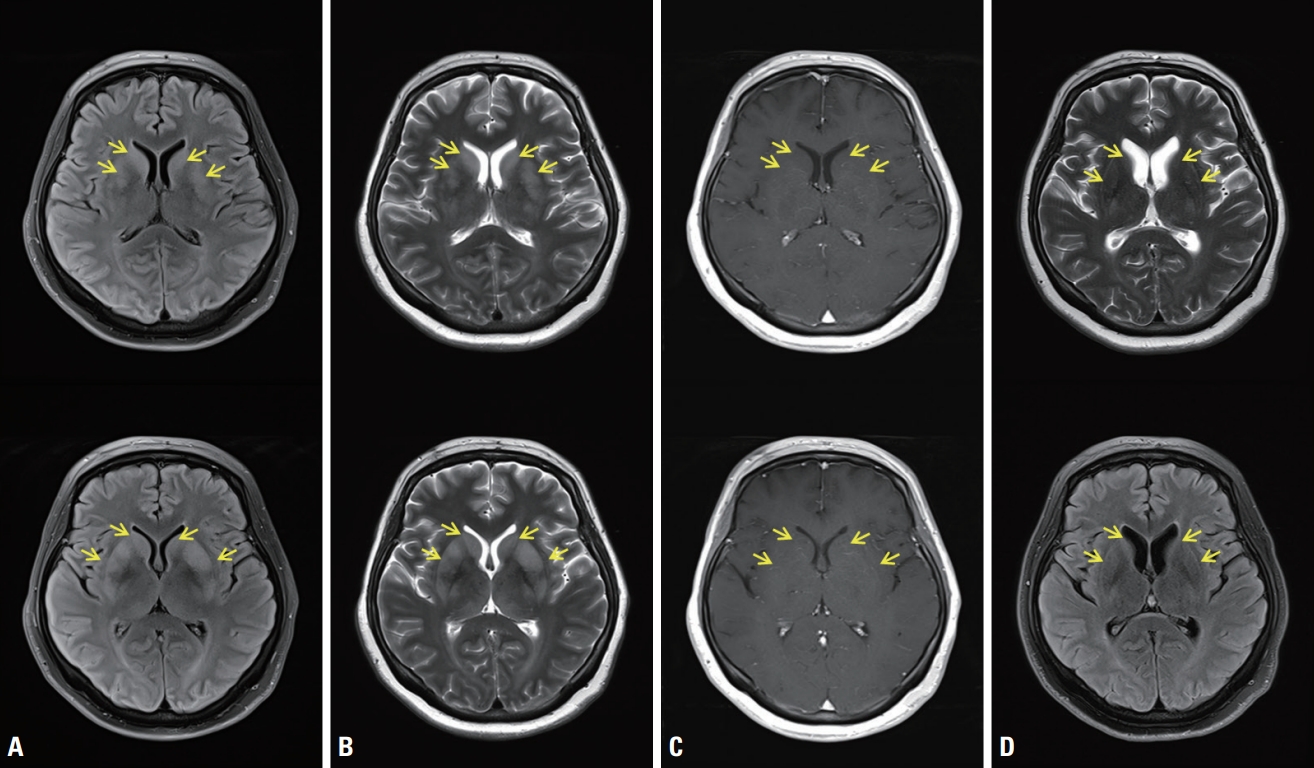
Fig. 2.MRI of the conus medullaris showed thickened nerve roots (orange arrows) in axial T2WI (A) and their gadolinium contrast enhancement (B). 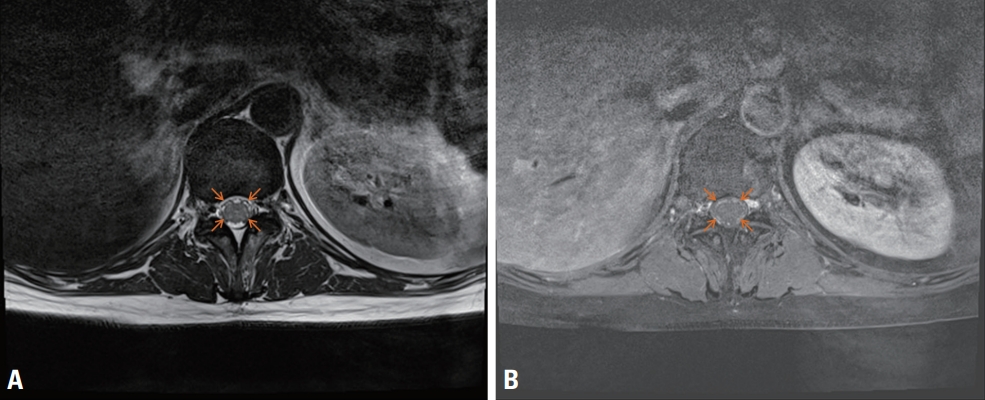
Table 1.Results of motor nerve conduction studies (NCSs)
The first NCSs performed on day 7 after symptom onset are normal except mildly slowed left ulnar motor nerve conduction velocity (CV) and absent H-reflex bilaterally, suggestive of early Guillain-Barré syndrome. Follow-up NCSs performed on day 21 (2 weeks after 1st NCSs reveal markedly reduced compound muscle action potential (CMAP) in both peroneal and both tibial nerves and persistently absent H-reflex. Mildly reduced CMAP in both median and both ulnar nerves are also observed. In contrast, results of sensory NCSs are completely normal in successive two tests (Supplementary Table 1). No parameters exceeding demyelinating criteria are observed such as prolonged motor distal latency 150% above upper limit of normal, slowing CV 70% less than lower limit of normal, motor conduction block or/and abnormal temporal dispersion. Taken together, these results are compatible to acute motor axonal neuropathy. DL, distal latency; CV, conduction velocity; RT, right; LT, left; M, motor; Ref., reference range; BE, below the elbow; AE, above the elbow; FH, fibula head; NR, no response. REFERENCES1. Ellul MA, Benjamin L, Singh B, Lant S, Michael BD, Easton A, et al. Neurological associations of COVID-19. Lancet Neurol 2020;19:767-783.
2. Palaiodimou L, Stefanou MI, Katsanos AH, Fragkou PC, Papadopoulou M, Moschovos C, et al. Prevalence, clinical characteristics and outcomes of Guillain-Barré syndrome spectrum associated with COVID-19: a systematic review and meta-analysis. Eur J Neurol 2021;28:3517-3529.
3. Siow I, Lee KS, Zhang JJY, Saffari SE, Ng A. Encephalitis as a neurological complication of COVID-19: a systematic review and meta-analysis of incidence, outcomes, and predictors. Eur J Neurol 2021;28:3491-3502.
4. Bertrand A, Leclercq D, Martinez-Almoyna L, Girard N, Stahl JP, De-Broucker T. MR imaging of adult acute infectious encephalitis. Med Mal Infect 2017;47:195-205.
5. Pohl D, Alper G, Van Haren K, Kornberg AJ, Lucchinetti CF, Tenembaum S, et al. Acute disseminated encephalomyelitis: updates on an inflammatory CNS syndrome. Neurology 2016;87(9 Suppl 2): S38-S45.
6. Gulko E, Oleksk ML, Gomes W, Ali S, Mehta H, Overby P, et al. MRI brain findings in 126 patients with COVID-19: initial observations from a descriptive literature review. AJNR Am J Neuroradiol 2020;41:2199-2203.
7. Van Cauter S, Severino M, Ammendola R, Van Berkel B, Vavro H, van den Hauwe L, et al. Bilateral lesions of the basal ganglia and thalami (central grey matter)-pictorial review. Neuroradiology 2020;62:1565-1605.
8. Werner R, Lohr B, Lodemann P, Chemnitz JM, Woehrle JC. Autoimmune basal ganglia encephalitis with hemolytic anemia. Ann Hematol 2022;101:1857-1858.
9. Pawela C, Brunsdon RK, Williams TA, Porter M, Dale RC, Mohammad SS. The neuropsychological profile of children with basal ganglia encephalitis: a case series. Dev Med Child Neurol 2017;59:445-448.
10. Fokke C, van den Berg B, Drenthen J, Walgaard C, van Doorn PA, Jacobs BC. Diagnosis of Guillain-Barré syndrome and validation of Brighton criteria. Brain 2014;137(Pt 1): 33-43.
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||