
|
| About the Journal |
| Aims and Scope |
| Journal Information |
| Editorial Board |
| Best Practice |
| Subscriptions |
| Contact Us |


|
| About the Journal |
| Aims and Scope |
| Journal Information |
| Editorial Board |
| Best Practice |
| Subscriptions |
| Contact Us |

AbstractComplex regional pain syndrome (CRPS) is a chronic regional pain disorder that most frequently affects the limbs. It is characterized by hyperalgesia, allodynia, edema, motor disturbance, and vasomotor instability, and typically occurs following surgery or trauma. In type-I CRPS there is no confirmed nerve injury, while peripheral nerve injury is present in type-II CRPS. The multifactorial pathophysiological etiology of CRPS includes inflammation, autoimmune responses, abnormal cytokine production, autonomic dysfunction, altered blood flow, psychological factors, and central cortical reorganization. There are no specific laboratory diagnostic tools for CRPS, and so it is diagnosed clinically. The Budapest criteria are currently the most-accepted diagnostic criteria.
INTRODUCTIONComplex regional pain syndrome (CRPS) is a chronic pain disorder that frequently causes allodynia (disproportionately increased pain response to a nonnoxious stimulus) and hyperalgesia (disproportionately increased pain response to a mildly noxious stimulus) in the extremities and other body regions. It occurs most frequently after trauma or surgery, and the pain experienced is excessive relative to the severity of the event and the amount of tissue damage.1 Clinical manifestations include autonomic dysfunction, hyperalgesia, edema, vasomotor instability, and spontaneous pain. Changes in the understanding of the illness over time have made diagnosing and managing CRPS challenging. Furthermore, a CRPS diagnosis cannot be confirmed by a single test, and so clinical diagnostic criteria for CRPS have been developed by clinicians and researchers based on the existing knowledge in the absence of information on the precise cause of CRPS. The clinical presentation and disease course of CRPS both vary. Despite receiving treatment, many patients have a poor prognosis, and their quality of life can be severely impacted. Reducing disease progression and improving the quality of life require early diagnosis and initiation of treatment.2 This review provides an update on the current literature related to the epidemiology, pathophysiology, clinical manifestations, and diagnosis of CRPS.
DEFINITION AND TERMINOLOGYThe definition of and diagnosis methods for CRPS have evolved over time. There are now more than 100 names for this disorder in the literature, depending on the triggering event, the predominating symptoms, the specialty and country of origin of the treating physician, and the hypothesized pathogenic mechanism.3 “Reflex sympathetic dystrophy,” “causalgia,” “algoneurodystrophy,” and “Sudeck’s atrophy” were historically used as names for this syndrome (Table 1),4 with “reflex sympathetic dystrophy” (RSD) being the most frequently accepted one. The term RSD was introduced by Evans in 1946 in an attempt to unify both causalgia and similar entities being due to a hyperactive sympathetic nervous system.5 The general consensus regarding CRPS has been reported as follows: “CRPS describes an array of painful conditions that are characterized by continuing (spontaneous and/or evoked) regional pain that is seemingly disproportionate in time or degree to the usual course of any known trauma or other lesion. The pain is regional (not in a specific nerve territory or dermatome) and usually has a distal predominance of abnormal sensory, motor, sudomotor, vasomotor, and/or trophic findings. The syndrome shows variable progression over time.”6 CRPS can be classified into two categories: (1) type-I CRPS (CRPS-I, the form also known as RSD), which is when there is no confirmed nerve injury, and (2) type-II CRPS (CRPS-II, formerly termed “causalgia”), which is associated with nerve injury.1 Some researchers additionally distinguish between “warm” and “cold” CRPS subtypes:7 increased skin temperature at outset of symptoms distinguishes warm CRPS, further indicating that it is an inflammatory form, while decreased skin temperature at the onset of symptoms distinguishes cold CRPS.
EPIDEMIOLOGYThere are obviously variations among practitioners because CRPS is diagnosed clinically based on the medical history, and physical and neurological examinations. This has resulted in the estimated annual incidence varying significantly between studies, from 5 to 26 per 100,000.8,9 The largest CRPS population-based study to date in the USA was reported on in 2016. Among the 33,406,123 patients included in that study from a sample database spanning from 2007 to 2011 revealed, 22,533 patients (i.e., 0.07%) were discharged with a diagnosis of CRPS.10 CRPS-I is more common than CRPS-II. The reported incidence of CRPS-II in the presence of peripheral nerve damage in different data sets has ranged from 2% to 14%, with a mean of approximately 4%.8
The reported female-to-male ratio of patients with CRPS has ranged from 2:1 to 4:1.9,11 The incidence appears to be highest in females who are postmenopausal.12 It is particularly interesting that a slight male preponderance in the sex distribution was found in two different Korean patient groups,13 while females made up nearly two-thirds of the patients in a Japanese cohort.14 The prevalence of CRPS varies with age, with the mean age at diagnosis peaking at 40-50 years and the incidence peaking at 61-70 years, and CRPS being very uncommon in childhood and adolescence.9
Polytrauma, fractures, crush injuries, and surgery are the most commonly recognized risk factors for CRPS.6,15 However, in up to 10% of patients there are no known triggering factors.9,11,12 The most frequent primary causes of CRPS in a recent review of 1,043 patients were fractures (42%), blunt traumatic injuries excluding fractures (e.g., sprains), surgery (12%), and carpal tunnel syndrome (7%).11 CRPS-I was found significantly more often in patients with ankle fractures than in those with hand, wrist, or foot fractures.16 Significantly higher incidence rates of CRPS-I were also related to intra-articular fractures and dislocated fractures.16 Nevertheless, the overall prevalence of CRPS associated with these triggering events is quite low. Crijns et al.17 found that 0.19% of 59,765 patients received treatment for distal radius fractures also had CRPS. In a similar more-recent study, 0.64% of 172,194 patients with distal radius fractures who underwent surgical treatment experienced CRPS.18 Among 113 cases that were analyzed retrospectively, the symptoms manifested on the right side in 47% of cases, on the left in 51%, and bilaterally in 2%.9,14 The upper extremities are more likely than the lower extremities to be affected.19 Two Korean cohorts represent a significant exception, where a lower limb was affected more frequently than an upper extremity, and 7% of patients had multiple extremities affected.20
A diagnosis of CRPS is strongly and independently correlated with fibromyalgia, which increases the risk up to 2.5-fold compared with controls.17,21 The risk may also be increased by other musculoskeletal diseases, including rheumatoid arthritis.19 An unexpectedly finding is that lower rates of CRPS-1 were related to diabetes, obesity, and hypothyroidism.15
PATHOPHYSIOLOGYWhile the pathophysiology of CRPS is still unknown, it is considered to involve dysfunction of both the central and peripheral nerve systems (Fig. 1).22 Despite scientific progress, the pathophysiological etiology of CRPS has still not been established (Table 2). CRPS is characterized by an inappropriate tissue response to injury as well as increased sensitization of the peripheral and central nervous systems along with accompanying inflammatory alterations and autonomic dysfunction.23 It is also thought that genetic and psychological factors contribute of CRPS.23
The warm or acute phase is defined by the release of proinflammatory modulators, while the cold or chronic phase is characterized by the activation of keratinocytes, fibroblasts, and osteoclasts.2 Both a proinflammatory immune response and impaired neuropeptide signaling are characteristics of CRPS.24 Inflammation is a normal natural consequence of a stroke, surgery, or tissue trauma, and the response of the innate immune system is both amplified and persistent in CRPS patients.2 Cytokines of the innate immune system trigger an immune cascade that results in histamine-induced vasodilation, which in turn results in the redness, swelling, pain, and warmth typical of the acute phase of CRPS.25 Numerous investigations have found increased CD4 and CD8 lymphocyte populations in CRPS patients, suggesting an antigen-mediated T-cell response.26 Neuropathic inflammation is also considered a major factor in the progression of CRPS. The activation of peripheral nociceptors of C-fibers results in the transmission of pain signals afferently toward the dorsal ganglia and efferently toward the affected tissue.27 An investigation of skin biopsies from CRPS patients found that the binding of substance P and calcitonin-gene-related peptide (CGRP) to keratinocyte receptors led to keratinocyte proliferation, neurogenic inflammatory alterations, and subsequent hyperalgesia and allodynia.28 It was believed that depolarization of afferent C-fibers could lead in neurogenic inflammation and the release of substance P and CGRP.29
Previous research has found that autoantibody levels in the serum, skin, and tissues are higher in CRPS patients and animal models, which supports CRPS being an autoimmune disorder.30 Autoantibodies are thought to cause pain in CRPS by sensitizing nociceptors.31 Additionally, CRPS induced elevations of IgM antibody levels in the skin and spinal tissue of rats, which were assumed to cause increased nociceptive sensitization.32
Abnormal activity of the autonomic nervous system was initially believed to be the cause of CRPS. Clinical symptoms such as skin-color changes, elevated heart rate, decreased heart-rate variability, low cardiac output, and excessive sweating are frequently caused by an imbalance in the autonomic nervous system in CRPS patients.33
Postganglionic sympathetic neurons and afferent neurons are connected via the sympathetic nervous system, and so it is thought that upregulation of alpha-adrenergic receptors causes inflammation of the skin of CRPS patients.34 Patients with CRPS were found to have higher systemic catecholamine expression but lower norepinephrine levels in the affected limb.34 Prolonged proinflammatory cytokine release during the chronic cold phase of CRPS causes hyperactivity of the sympathetic nervous system, which increases norepinephrine levels, decreases α-1 adrenergic receptor expression, and culminates in vasoconstriction and the development of a cold, blue, and clammy limb.2
Another suggested underlying mechanism is abnormal cortical reorganization, which has also been described as a form of maladaptive plasticity. Studies utilizing functional magnetic resonance imaging (MRI) and magnetoencephalography have revealed this brain reorganization.35 Somatosensory cortex reorganization may cause various CRPS manifestations, such as the glove- or stocking-like distribution of sensory signs,36 the extension of sensory deficits over an entire quadrant or even one entire side of the body,37 referral or mislocalization of tactile sensations,38 and the perception of the affected limb as being distorted or larger than it actually is.39 Similar to this, reorganization of the motor cortex definitely plays a significant role in the majority of motor abnormalities experienced by patients with CRPS-I or CRPS-II.40
There is abundant evidence that hereditary factors contribute to the propensity to develop CRPS. There have been reports of familial cases of CRPS, and these patients frequently have severe disease with early onset, recurrences, spreading to many sites, and/or dystonia.41 HLA-DQ1 was substantially more prevalent in 52 Dutch CRPS patients than in 295 local controls.42 A pilot study produced indirect relationship between CRPS and the closely linked DR2.43 CRPS manifestations are potentially related to genetic abnormalities in mitochondrial DNA.44
Psychosocial factors and pain outcomes are correlated in CRPS patients.45 It has been proposed that psychological conditions including depression play a role in the emergence of CRPS.4 Most (86%) of 58 CRPS patients who met the criteria for posttraumatic stress disorder (PTSD) had symptoms of PTSD before being diagnosed as CRPS, with the remaining 14% of patients developing PTSD during the course of CRPS.46 However, research findings on how psychological factors contribute to the development of CRPS are controversial.
CLINICAL MANIFESTATIONSThe main characteristics of CRPS include spontaneous pain, hyperalgesia, allodynia, and abnormal vasomotor and sudomotor activities.36 Psychological dysfunction frequently manifests during the course of CRPS. The signs and symptoms of CRPS can vary widely between patients and over the course of the disease (Table 3).47
Most adult patients diagnosed with CRPS-I or CRPS-II exhibit a combination of sensory gain and loss.48 Patients with CRPS may complain of localized, intense somatic tissue pain that is burning, throbbing, squeezing, aching, or shooting.49 The pain typically progresses from the site of the initial injury, and may affect the entire limb in its most-extreme manifestation, or even the limb on the opposite side in rare cases.50 One-third of CRPS patients have severe allodynia, which is more common in the chronic stage and is considered to be caused by central nociceptive sensitization.51 Patients who have allodynia frequently adopt a protective posture to safeguard the injured extremity. Many CRPS patients are unable to use the affected extremity due to movement exacerbating the pain. CRPS-related pain does not have a dermatomal or nerve root distribution, and typically is not related to a distinct nerve lesion. Most CRPS patients experience some type of motor dysfunction,36,52,53 such as tremor, restricted range of motion, muscle weakness, or the affected limb being fixed in a long-term position (dystonia).52 Additionally, CRPS patients frequently move more slowly or more inaccurately when performing specific movements with the affected limb.54-56 Trophic changes such as osteoporosis, fibrosis, thin or shiny skin, aberrant nail growth, and changes in hair growth may be present, especially in the chronic stage. Sudomotor dysfunction (hyperhidrosis or hypohidrosis) can also occur in CRPS patients.36,48 Vasomotor or sudomotor dysfunction is a sign of an unstable sympathetic nervous system. Significant changes in skin temperature and (in particular) color occur over time and are not constant manifestations,57 with the limb initially having a dry, hot, and pink appearance in the classic presentation, but later becoming blue, cold, and hyperhydrotic.58 Usually the skin temperature differs by more than 1°C between the affected and unaffected limbs.59 Regardless of the skin color or temperature, edema is observable in almost all patients at the initiation of symptoms.36 This occurs less frequently as the disease becomes present for longer, but at least 40% of patients with chronic diseases still experience edema.51
It has long been believed that CRPS develops in three separate phases: (1) an acute phase characterized by the predominance of pain, sensory abnormalities, edema, sudomotor dysfunction, and possible motor function symptoms; (2) a dystrophic phase when trophic and substantial motor alterations arise, pain and sensory dysfunction become more pronounced, and vasomotor abnormalities persist; and (3) an atrophic phase where motor and trophic changes progress, vasomotor disturbances persist, and pain and sensory problems lessen.60
DIAGNOSTIC CRITERIAReflex sympathetic dystrophy was first identified using the Veldman criteria in 1993 (Table 4).36 There are no specific tools or equipment required to fulfill these criteria. The International Association for the Study of Pain (IASP) published the first CRPS diagnostic criteria in 1994. These criteria later became known as the Orlando criteria (Table 4),61 and the subsequent establishment of the Budapest criteria followed further work by a consensus group.6
The Budapest Research Criteria were established by Harden et al.,62 and were subsequently adopted as new criteria for diagnosing CRPS. Sensory abnormalities, temperature asymmetry, color changes, edema, sweating, and motor dysfunction were the most important contributing factors. These criteria closely reflected criteria proposed by Harden and Bruehl in 1999.63,64 A consensus meeting was convened by the IASP in 2003 with the goal of improving the IASP diagnostic criteria for CRPS, which yielded new criteria known as the Budapest criteria.6 The Budapest criteria are currently the most widely used diagnostic criteria since they have recently been validated as having higher specificity than the earlier criteria (Table 4).15 The Budapest criteria were compared with the IASP criteria in order to distinguish between CRPS-I and other types of neuropathic pain during the initial validation of these criteria in 2010.15 That study found excellent diagnostic sensitivity for both the IASP criteria (100%) and the Budapest criteria (99%), but the specificity was higher for the Budapest criteria (68%) than for the IASP criteria (41%).15 The diagnostic validity of the Budapest criteria has been assessed in four studies, which have produced varying results, with the sensitivity ranging from 45% to 99% and the specificity ranging from 68% to 85%.11,14,64 Despite broad agreement on its application, the Budapest criteria for clinically diagnosing CRPS-I and CRPS-II might not be applicable to all populations or all neurological disorders, including poststroke CRPS.64
EVALUATIONThere are no specific laboratory diagnostic tools for CRPS, and so it is diagnosed clinically. However, various diagnostic tools are frequently used to support the diagnosis of CRPS (Table 5). Additionally, several tests are used to evaluate autonomic function and aid in excluding other diseases. Careful physical and neurological examinations should be applied to patients with suspected CRPS. To help rule out infection and rheumatological problems, general laboratory tests such a full blood count, C-reactive protein, erythrocyte sedimentation rate, and serum autoantibodies are recommended. To rule out nerve lesions, nerve conduction velocity tests and electromyography should be performed. Somatosensory evoked potential studies and transcranial magnetic stimulation of motor pathways should be additionally used to assess central pathways. Bedside sensory testing or even quantitative sensory testing may be useful for identifying small-fiber impairment. These include the quantitative sudomotor axon reflex test (QSART), thermography, and laser Doppler flowmetry for testing sudomotor function and peripheral vasoconstrictor responses. The use of thermography is predicated on the existence of unilateral vasomotor disruption, which causes detectable temperature differences between limbs.36 However, normal symmetrical thermographic findings do not rule out a CRPS diagnosis. In both the acute and chronic stages of the disease, quantitative analysis of sudomotor activity may reveal abnormal sweat production of the affected extremity.65 A provocative test called the QSART measures the amount of sweat produced in response to a cholinergic challenge, such as acetylcholine.
Numerous imaging methods including plain radiography, bone scintigraphy, computed tomography, MRI, positron-emission tomography, and single-photon-emission computed tomography are the most beneficial for ruling out alternative diagnoses. Although three-phase bone scintigraphy has long been regarded as an objective diagnosis method, it is insufficient for diagnosing CRPS.66 However, CRPS is considered to be characterized by a pattern of increased uptake in all three phases, particularly diffuse periarticular uptake in and around the joints of the affected extremity during the delayed phase, especially during the first 6 months of CRPS.66 Identifying patients who might benefit from bisphosphonate therapy may require the use of bone scan.67,68 Additionally, MRI can reveal periarticular bone-marrow edema, soft-tissue swelling, joint effusions, and atrophy of periarticular structures in later stages.69 In a subgroup of CRPS patients, sympathetically maintained pain can be identifying by applying a diagnostic block of sympathetic nerves using local anesthetic.70 A more-permanent sympathetic block can then be achieved percutaneously using phenol or radiofrequency ablation if sympathetic blockade significantly reduces pain.71,72
CONCLUSIONCRPS is a complex disorder that is notoriously difficult to treat and frequently manifests with permanent debilitating sequelae. There is no single standard diagnostic test to confirm the diagnosis, but these patients can be identified using well-established clinical criteria. Although there have been considerable advances since the early definitions, the current knowledge of CRPS remains insufficient. A better understanding of the mechanisms underlying this disorder requires larger clinical investigations of higher quality. This will lead to faster diagnoses and the development of more precisely targeted treatments.
Fig. 1.Hypothetical model of mechanisms interacting in the development of complex regional pain syndrome. PTSD, post-traumatic stress disorder; CGRP, calcitonin-gene-related peptide; IL, interleukin; TNF, tumor necrosis factor. 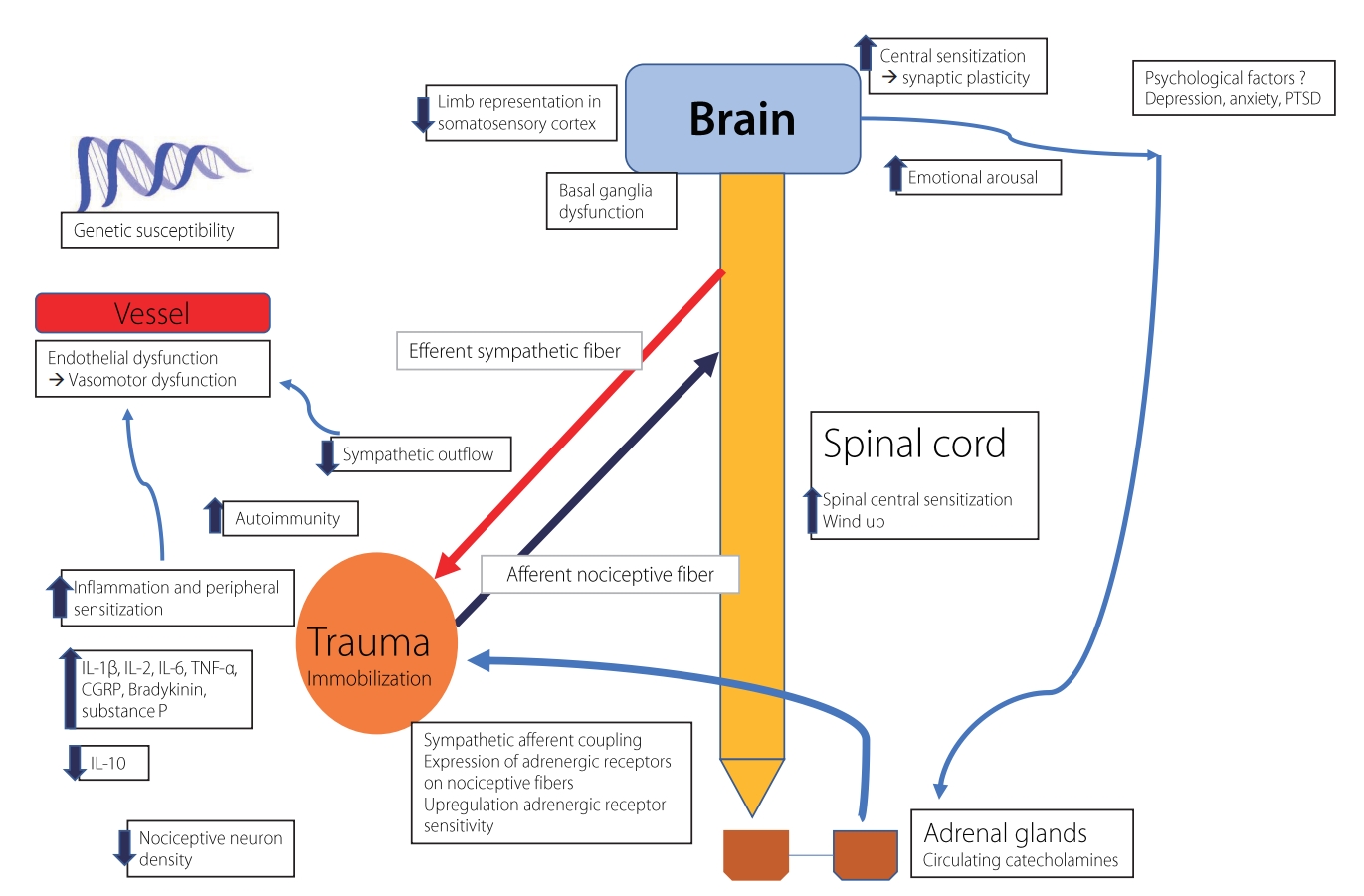
Table 1.Synonyms for CRPS Table 2.The various pathophysiologies of CRPS Table 3.Signs and symptoms of CRPS Table 4.Criteria for diagnosing CRPS Table 5.Diagnostic tests for CRPS Table 6.Differential diagnosis REFERENCES1. Treede RD, Rief W, Barke A, Aziz Q, Bennett MI, Benoliel R, et al. Chronic pain as a symptom or a disease: the IASP classification of chronic pain for the international classification of diseases (ICD-11). Pain 2019;160:19-27.
2. Misidou C, Papagoras C. Complex regional pain syndrome: an update. Mediterr J Rheumatol 2019;30:16-25.
3. Todorova J, Dantchev N, Petrova G. Complex regional pain syndrome acceptance and the alternative denominations in the medical literature. Med Princ Pract 2013;22:295-300.
4. Eldufani J, Elahmer N, Blaise G. A medical mystery of complex regional pain syndrome. Heliyon 2020;6:e03329.
5. Evans JH. Notes on the folklore and legends associated with the Kentish megaliths. Folklore 1946;57:36-43.
6. Harden RN, Bruehl S, Stanton-Hicks M, Wilson PR. Proposed new diagnostic criteria for complex regional pain syndrome. Pain Med 2007;8:326-331.
7. Eberle T, Doganci B, Krämer HH, Geber C, Fechir M, Magerl W, et al. Warm and cold complex regional pain syndromes: differences beyond skin temperature? Neurology 2009;72:505-512.
8. Moseley GL. Graded motor imagery for pathologic pain: a randomized controlled trial. Neurology 2006;67:2129-2134.
9. Sandroni P, Benrud-Larson LM, McClelland RL, Low PA. Complex regional pain syndrome type I: incidence and prevalence in Olmsted county, a population-based study. Pain 2003;103:199-207.
10. Elsharydah A, Loo NH, Minhajuddin A, Kandil ES. Complex regional pain syndrome type 1 predictors-epidemiological perspective from a national database analysis. J Clin Anesth 2017;39:34-37.
11. Ott S, Maihöfner C. Signs and symptoms in 1,043 patients with complex regional pain syndrome. J Pain 2018;19:599-611.
12. de Mos M, de Bruijn AG, Huygen FJ, Dieleman JP, Stricker BH, Sturkenboom MC. The incidence of complex regional pain syndrome: a population-based study. Pain 2007;129:12-20.
13. Kang JE, Kim YC, Lee SC, Kim JH. Relationship between complex regional pain syndrome and working life: a Korean study. J Korean Med Sci 2012;27:929-933.
14. Sumitani M, Shibata M, Sakaue G, Mashimo T. Development of comprehensive diagnostic criteria for complex regional pain syndrome in the Japanese population. Pain 2010;150:243-249.
15. Harden NR, Bruehl S, Perez RSGM, Birklein F, Marinus J, Maihofner C, et al. Validation of proposed diagnostic criteria (the “Budapest Criteria”) for complex regional pain syndrome. Pain 2010;150:268-274.
16. Beerthuizen A, Stronks DL, Van’t Spijker A, Yaksh A, Hanraets BM, Klein J, et al. Demographic and medical parameters in the development of complex regional pain syndrome type 1 (CRPS1): prospective study on 596 patients with a fracture. Pain 2012;153:1187-1192.
17. Crijns TJ, van der Gronde BATD, Ring D, Leung N. Complex regional pain syndrome after distal radius fracture is uncommon and is often associated with fibromyalgia. Clin Orthop Relat Res 2018;476:744-750.
18. Jo YH, Kim K, Lee BG, Kim JH, Lee CH, Lee KH. Incidence of and risk factors for complex regional pain syndrome type 1 after surgery for distal radius fractures: a population-based study. Sci Rep 2019;9:4871.
19. Littlejohn GO. Reflex sympathetic dystrophy in adolescents: lessons for adults. Arthritis Rheum 2004;51:151-153.
20. Choi YS, Lee MG, Lee HM, Lee CJ, Jo JY, Jeon SY, et al. Epidemiology of complex regional pain syndrome: a retrospective chart review of 150 Korean patients. J Korean Med Sci 2008;23:772-775.
21. van Velzen GAJ, Huygen FJPM, van Kleef M, van Eijs FV, Marinus J, van Hilten JJ. Sex matters in complex regional pain syndrome. Eur J Pain 2019;23:1108-1116.
22. Marinus J, Moseley GL, Birklein F, Baron R, Maihöfner C, Kingery WS, et al. Clinical features and pathophysiology of complex regional pain syndrome. Lancet Neurol 2011;10:637-648.
23. Shim H, Rose J, Halle S, Shekane P. Complex regional pain syndrome: a narrative review for the practising clinician. Br J Anaesth 2019;123:e424-e433.
24. Li WW, Guo TZ, Shi X, Birklein F, Schlereth T, Kingery WS, et al. Neuropeptide regulation of adaptive immunity in the tibia fracture model of complex regional pain syndrome. J Neuroinflammation 2018;15:105.
25. Dirckx M, Stronks DL, van Bodegraven-Hof EA, Wesseldijk F, Groeneweg JG, Huygen FJ. Inflammation in cold complex regional pain syndrome. Acta Anaesthesiol Scand 2015;59:733-739.
26. Russo MA, Georgius P, Pires AS, Heng B, Allwright M, Guennewig B, et al. Novel immune biomarkers in complex regional pain syndrome. J Neuroimmunol 2020;347:577330.
27. Littlejohn G. Neurogenic neuroinflammation in fibromyalgia and complex regional pain syndrome. Nat Rev Rheumatol 2015;11:639-648.
28. Birklein F, Drummond PD, Li W, Schlereth T, Albrecht N, Finch PM, et al. Activation of cutaneous immune responses in complex regional pain syndrome. J Pain 2014;15:485-495.
29. Gherardini G, Curinga G, Colella G, Freda N, Rauso R. Calcitonin gene-related peptide and thermal injury: review of literature. Eplasty 2009;9:e30.
31. Weber M, Birklein F, Neundörfer B, Schmelz M. Facilitated neurogenic inflammation in complex regional pain syndrome. Pain 2001;91:251-257.
32. van Velzen GA, Rombouts SA, van Buchem MA, Marinus J, van Hilten JJ. Is the brain of complex regional pain syndrome patients truly different? Eur J Pain 2016;20:1622-1633.
33. Knudsen LF, Terkelsen AJ, Drummond PD, Birklein F. Complex regional pain syndrome: a focus on the autonomic nervous system. Clin Auton Res 2019;29:457-467.
34. Jänig W, Levine JD, Michaelis M. Interactions of sympathetic and primary afferent neurons following nerve injury and tissue trauma. Prog Brain Res 1996;113:161-184.
35. Di Pietro F, McAuley JH, Parkitny L, Lotze M, Wand BM, Moseley GL, et al. Primary motor cortex function in complex regional pain syndrome: a systematic review and meta-analysis. J Pain 2013;14:1270-1288.
36. Veldman PH, Reynen HM, Arntz IE, Goris RJ. Signs and symptoms of reflex sympathetic dystrophy: prospective study of 829 patients. Lancet 1993;342:1012-1016.
37. Rommel O, Gehling M, Dertwinkel R, Witscher K, Zenz M, Malin JP, et al. Hemisensory impairment in patients with complex regional pain syndrome. Pain 1999;80:95-101.
38. Maihöfner C, Neundörfer B, Birklein F, Handwerker HO. Mislocalization of tactile stimulation in patients with complex regional pain syndrome. J Neurol 2006;253:772-779.
40. Galer BS, Butler S, Jensen MP. Case reports and hypothesis: a neglect-like syndrome may be responsible for the motor disturbance in reflex sympathetic dystrophy (complex regional pain syndrome-1). J Pain Symptom Manage 1995;10:385-391.
41. de Rooij AM, de Mos M, Sturkenboom MC, Marinus J, van den Maagdenberg AM, van Hilten JJ. Familial occurrence of complex regional pain syndrome. Eur J Pain 2009;13:171-177.
42. van de Beek WJ, van Hilten JJ, Roep BO. HLA-DQ1 associated with reflex sympathetic dystrophy. Neurology 2000;55:457-458.
43. Mailis A, Wade J. Profile of Caucasian women with possible genetic predisposition to reflex sympathetic dystrophy: a pilot study. Clin J Pain 1994;10:210-217.
44. Higashimoto T, Baldwin EE, Gold JI, Boles RG. Reflex sympathetic dystrophy: complex regional pain syndrome type I in children with mitochondrial disease and maternal inheritance. Arch Dis Child 2008;93:390-397.
45. Park HY, Jang YE, Oh S, Lee PB. Psychological characteristics in patients with chronic complex regional pain syndrome: comparisons with patients with major depressive disorder and other types of chronic pain. J Pain Res 2020;13:389-398.
46. Speck V, Schlereth T, Birklein F, Maihöfner C. Increased prevalence of posttraumatic stress disorder in CRPS. Eur J Pain 2017;21:466-473.
47. Bussa M, Guttilla D, Lucia M, Mascaro A, Rinaldi S. Complex regional pain syndrome type I: a comprehensive review. Acta Anaesthesiol Scand 2015;59:685-697.
48. Gierthmühlen J, Maier C, Baron R, Tölle T, Treede RD, Birbaumer N, et al. Sensory signs in complex regional pain syndrome and peripheral nerve injury. Pain 2012;153:765-774.
49. Raja SN, Grabow TS. Complex regional pain syndrome I (reflex sympathetic dystrophy). Anesthesiology 2002;96:1254-1260.
50. Karacan I, Aydin T, Ozaras N. Bone loss in the contralateral asymptomatic hand in patients with complex regional pain syndrome type 1. J Bone Miner Metab 2004;22:44-47.
51. Birklein F, Riedl B, Sieweke N, Weber M, Neundörfer B. Neurological findings in complex regional pain syndromes--analysis of 145 cases. Acta Neurol Scand 2000;101:262-269.
52. de Mos M, Huygen FJ, van der Hoeven-Borgman M, Dieleman JP, Ch Stricker BH, Sturkenboom MC. Outcome of the complex regional pain syndrome. Clin J Pain 2009;25:590-597.
53. de Boer RD, Marinus J, van Hilten JJ, Huygen FJ, van Eijs F, van Kleef M, et al. Distribution of signs and symptoms of complex regional pain syndrome type I in patients meeting the diagnostic criteria of the International Association for the Study of Pain. Eur J Pain 2011;15:830.e1-e8.
54. Schilder JCM, Schouten AC, Perez RSGM, Huygen FJPM, Dahan A, Noldus LPJJ, et al. Motor control in complex regional pain syndrome: a kinematic analysis. Pain 2012;153:805-812.
55. Uçeyler N, Eberle T, Rolke R, Birklein F, Sommer C. Differential expression patterns of cytokines in complex regional pain syndrome. Pain 2007;132:195-205.
56. Roganovic Z, Mandic-Gajic G. Pain syndromes after missile-caused peripheral nerve lesions: part 1--clinical characteristics. Neurosurgery 2006;59:1226. -1236. discussion 1236-1237.
57. Sherman RA, Karstetter KW, Damiano M, Evans CB. Stability of temperature asymmetries in reflex sympathetic dystrophy over time and changes in pain. Clin J Pain 1994;10:71-77.
58. Birklein F, Riedl B, Neundörfer B, Handwerker HO. Sympathetic vasoconstrictor reflex pattern in patients with complex regional pain syndrome. Pain 1998;75:93-100.
59. Wasner G, Schattschneider J, Heckmann K, Maier C, Baron R. Vascular abnormalities in reflex sympathetic dystrophy (CRPS I): mechanisms and diagnostic value. Brain 2001;124(Pt 3): 587-599.
60. Bruehl S, Harden RN, Galer BS, Saltz S, Backonja M, Stanton-Hicks M. Complex regional pain syndrome: are there distinct subtypes and sequential stages of the syndrome? Pain 2002;95(1-2): 119-124.
61. Reinders MF, Geertzen JH, Dijkstra PU. Complex regional pain syndrome type I: use of the International Association for the Study of Pain diagnostic criteria defined in 1994. Clin J Pain 2002;18:207-215.
62. Harden RN, Bruehl S, Galer BS, Saltz S, Bertram M, Backonja M, et al. Complex regional pain syndrome: are the IASP diagnostic criteria valid and sufficiently comprehensive? Pain 1999;83:211-219.
63. Bruehl S, Harden RN, Galer BS, Saltz S, Bertram M, Backonja M, et al. External validation of IASP diagnostic criteria for complex regional pain syndrome and proposed research diagnostic criteria. International Association for the Study of Pain. Pain 1999;81:147-154.
64. Oh SW, Choi SU, Park M, Shin JH. Validity of the Budapest criteria for poststroke complex regional pain syndrome. Clin J Pain 2019;35:831-835.
65. Birklein F, Sittl R, Spitzer A, Claus D, Neundörfer B, Handwerker HO. Sudomotor function in sympathetic reflex dystrophy. Pain 1997;69:49-54.
66. Ringer R, Wertli M, Bachmann LM, Buck FM, Brunner F. Concordance of qualitative bone scintigraphy results with presence of clinical complex regional pain syndrome 1: meta-analysis of test accuracy studies. Eur J Pain 2012;16:1347-1356.
67. Goldsmith DP, Vivino FB, Eichenfield AH, Athreya BH, Heyman S. Nuclear imaging and clinical features of childhood reflex neurovascular dystrophy: comparison with adults. Arthritis Rheum 1989;32:480-485.
68. Turner-Stokes L, Goebel A. Complex regional pain syndrome in adults: concise guidance. Clin Med (Lond) 2011;11:596-600.
69. Sintzoff S, Sintzoff S Jr, Stallenberg B, Matos C. Imaging in reflex sympathetic dystrophy. Hand Clin 1997;13:431-442.
70. Price DD, Long S, Wilsey B, Rafii A. Analysis of peak magnitude and duration of analgesia produced by local anesthetics injected into sympathetic ganglia of complex regional pain syndrome patients. Clin J Pain 1998;14:216-226.
71. Manchikanti L. The role of radiofrequency in the management of complex regional pain syndrome. Curr Rev Pain 2000;4:437-444.
72. Furlan AD, Lui PW, Mailis A. Chemical sympathectomy for neuropathic pain: does it work? Case report and systematic literature review. Clin J Pain 2001;17:327-336.
|
|
|||||||||||||||||||||||||||||||||||||