Transcranial magnetic stimulation parameters as neurophysiological biomarkers in Alzheimer’s disease
Article information
Abstract
Transcranial magnetic stimulation (TMS) is a safe and noninvasive tool for investigating the cortical excitability of the human brain and the neurophysiological functions of GABAergic, glutamatergic, and cholinergic neural circuits. Neurophysiological biomarkers based on TMS parameters can provide information on the pathophysiology of dementia, and be used to diagnose Alzheimer’s disease and differentiate different types of dementia. This review introduces the basic principles of TMS, TMS devices and stimulating paradigms, several neurophysiological measurements, and the clinical implications of TMS for Alzheimer’s disease.
INTRODUCTION
Transcranial magnetic stimulation (TMS) is a safe and non-invasive intervention for electrically stimulating neural tissues using magnetic pulses. After Barker et al.1 reported that the method could be used to safely stimulate the human brain in 1985, Pascual-Leone et al.2 reported that TMS could safely modulate brain activities by applying high-frequency repetitive TMS (rTMS). TMS has been used to investigate cortical excitability and provide additional evidence for use in diagnoses and prognoses. In addition, rTMS has been applied in therapeutic interventions for various neurological and psychiatric diseases. TMS has recently been introduced as a novel approach to diagnosing and differentiating dementia, and to enhance cognitive function in both cognitively normal and impaired patients.3 This article reviews the TMS measurements that provide information about the neurophysiological state of Alzheimer’s disease (AD) and the future value of TMS in the diagnosis and treatment of AD.
PRINCIPLES OF TMS
Action mechanism of TMS
TMS is based on Faraday’s law of electromagnetic induction.4 Rapidly changing pulses of current passing through a coil positioned above the head generate magnetic fields that penetrate the scalp and skull to reach the brain and induce secondary electric currents in the cortex.5,6 These electric currents depolarize the neuron membranes either directly or indirectly. They can markedly influence changes in excitability from the regional cortex to the subcortex and distant areas that are connected to the stimulated area via neural pathways,7-10 with these changes ultimately affecting human cognition and behavior.11
rTMS can be used to modulate synaptic plasticity, especially long-term potentiation (LTP)12 and long-term depression (LTD).13 LTP is a phenomenon in which synapses are persistently strengthened, and it is an important mechanism of memory and learning at the neuron level. High-frequency rTMS and theta-burst stimulation (TBS) affect the expression levels of various receptors, including N-methyl-D-aspartate (NMDA) receptors,14 and neurotransmitters.15 High-frequency rTMS reduces the synaptic conduction threshold, leading to enhanced synaptic connections and synaptic plasticity.16 These structural and functional changes in synapses lead to the induction of LTP, which affects cognitive function.
TMS devices and stimulation paradigms
A TMS device consists of a coil and a stimulator. There are several types of coil: circular or round, figure-of-eight, double-cone, and H-coil. The amplitude of currents flowing in the coil controls the intensity of the induced magnetic field, and the type of coil determines where that field is focused. A figure-of-eight coil is the most widely used and provides more focal stimulation compared with a circular coil, for which the electric field is more widely distributed and provides bihemispheric stimulation.17 An H-coil stimulates a wide and deep area of the brain,18 while a double-cone coil stimulates deep and focal areas such as deep parts of the cerebellum19 or the leg motor area.20
The stimulator generates pulse waves of appropriate frequency, intensity, and number of pulses to influence the effects of TMS.21 Generally, high-frequency stimulation (5-25 Hz) induces cortical excitability while low-frequency stimulation (≤ 1 Hz) inhibits cortical activities.22 However, high-frequency stimulation at low intensities decreases cortical activity, while high-frequency stimulation at high intensities increases cortical excitability.23 TBS is a variant of rTMS that increases cortical excitability when applied intermittently and decreases cortical excitability when applied continuously.24
TMS OUTCOME MEASURES IN DEMENTIA
Single-pulse TMS
Single-pulse TMS is generally used to estimate the motor threshold (MT) or cortical excitability when recording motor evoked potentials (MEPs). When single-pulse TMS is applied to the primary motor cortex, series of recordable corticospinal volleys are generated that reflect the trans-synaptic activation of cortical neurons.
Motor threshold
The MT is related to the integrity of the corticospinal pathway. It is used to characterize each level of excitability along the pathway, or as a reference measure of excitability when determining the intensity to apply in different stimulation protocols. The resting MT (RMT) refers to the lowest TMS intensity that evokes MEPs with a peak-to-peak amplitude of ≥ 50 μV in at least 50% of successive trials in the relaxed target muscle.25
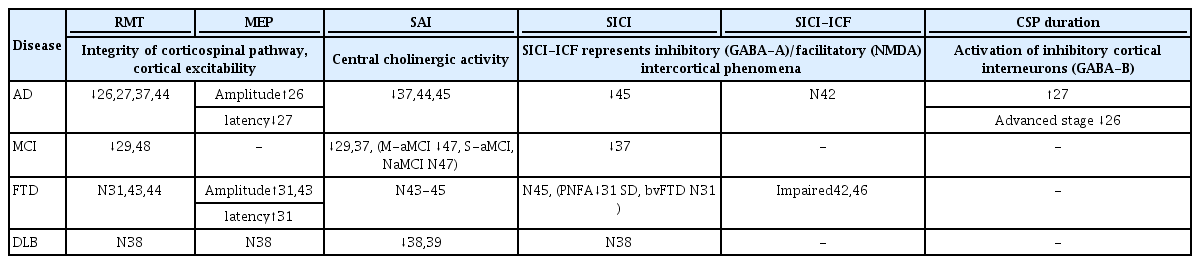
RMT is decreased in the early stage of AD and is lowest in the advanced stage.26 The reduced RMT in early-stage AD indicates hyperexcitability of the motor cortex, and a relative increase in excitability in the excitatory/inhibitory balance, which is caused by impairment of inhibitory interneuron function.27 Decreased RMT following disease progression might be compensatory for the loss of motor cortex neurons in the mild-to-moderate stage of AD.28 Mild cognitive impairment (MCI) patients also show a lower RMT than healthy subjects, similar to AD patients. These results suggest that the hyperexcitability emerges from the MCI state when the cognitive function is relatively good.29
Motor evoked potential
The MEP amplitude reflects the integrity of the corticospinal tract and the excitability of the motor cortex, nerve roots, and conduction along the peripheral motor pathway to the muscle. A reduced MEP amplitude reflects the failure of central motor conduction. The small-amplitude MEPs seen in stroke30 involve the motor cortex or corticospinal tract, hydrocephalus, and multiple sclerosis. Most studies have found that the MEP amplitude does not differ between AD patients and healthy controls, whereas MEPs are lower in frontotemporal dementia (FTD) patients. This indicates the involvement of the corticospinal tract in the early stage of FTD but not in AD.31
Contralateral silent period
Applying suprathreshold TMS to the motor cortex during tonic voluntary contraction of the contralateral muscles results in suppression of the electromyographic activity of those muscles lasting a few hundred milliseconds.32 This phenomenon is called the contralateral silent period (CSP), which reflects activation of the inhibitory cortical interneurons, which is mediated by gamma-aminobutyric acid (GABA)-B.33 Severe synapse loss with failure of inhibitory control represented as a significant reduction of the CSP in patients with advanced AD.34
Paired-pulse TMS
The paired-pulse TMS protocol involves the application of two different sequential stimuli with various interstimulus intervals (ISIs). This can provide information about the integrity of corticocortical connections. There are two inhibitory circuits and two facilitatory circuits determined by the intensity and ISI of this protocol: short-interval intracortical inhibition (SICI), long-interval intracortical inhibition (LICI), intracortical facilitation (ICF), and short-interval intracortical facilitation. Paired-pulse TMS, like short-latency afferent inhibition (SAI), is used to investigate sensory-motor interactions in the cerebral cortex.
SICI and ICF
A subthreshold magnetic conditioning stimulus (CS) is followed by a suprathreshold magnetic test stimulus (TS) delivered to the same target area using the same coil. The TS induces a corticospinal output leading to an MEP, while a subthreshold CS only excites local cortical interneurons. Therefore, this stimulation protocol can be used to assess the effects of interneurons on cortical output.35 Paired-pulse TMS shows inhibitory intercortical phenomena (with a short ISI of 1-4 ms; SICI) and facilitatory intercortical phenomena (with a longer ISI of > 5 ms; ICF) that are mediated by GABA-A and glutamatergic NMDA, respectively.36
Short-latency afferent inhibition
The SAI protocol involves applying conditioning electric stimulation to the median nerve at the wrist 20-25 ms before applying a contralateral TMS test pulse, with the timing compatible with the activation of the test pulse alone. SAI is a putative marker of central cholinergic activity and depends on the corticocortical inhibitory and thalamocortical circuits.
SICI and SAI are significantly decreased in patients with AD and MCI, which reflects low cholinergic activity.37 Other studies have found SAI to be normal in FTD patients, but reduced in patients with AD and dementia with Lewy bodies (DLB).38 DLB patients showed a significant reduction of cholinergic activity as measured by SAI, which was correlated with their hallucinations.39
Repetitive TMS
rTMS involves applying a train of single TMS pulses with a constant frequency and intensity for a given time to the brain target area. It can modulate cortical excitability, with low-frequency rTMS (≤ 1 Hz) suppressing cortical excitability40 and high-frequency rTMS (≥ 5 Hz) increasing cortical excitability.41 rTMS can also be used to noninvasively induce long-lasting modulation of the cortical excitability. The mechanism underlying how rTMS modulates cortical excitability beyond the duration of stimulation is unclear, but LTP and LTD of the cortical synapses are a possible mechanism. Such lasting modulation of cortical activity by rTMS can also be induced in areas outside the motor cortex, and these findings raise the possibility of therapeutic applications of rTMS.
CLINICAL APPLICATIONS IN DEMENTIA
Diagnostic and prognostic applications of TMS in dementia
Recent studies have suggested that TMS could be a novel tool for investigating the early features of synaptic dysfunction in AD patients and the identification of biomarkers.3,29,42 The initial AD pathological changes were considered to be the aggregation and accumulation of amyloid-beta and tau proteins that damaged neuron synapses long before the presentation of clinical dementia. These toxic oligomeric species impair synaptic transmission, which could be more precisely correlated with the severity of the disease and an additional cognitive decline related to AD.3 AD patients show different MTs and SAIs at different stages of the disease. A lower RMT, which reflects hyperexcitability of the motor cortex, presents in MCI and the early stage of AD. The RMT decreases with disease progression, and at an advanced stage, it reduces further with global cortical atrophy of the brain.26 Thus, TMS parameters well reflect the functional changes in synapses according to the state in AD patients.
The characteristics of TMS parameters differ among several types of dementia.43 In FTD, TMS has revealed central motor circuit abnormalities through reduced-amplitude or absent MEPs, and increased latency of MEPs and the central motor conduction time, even in the absence of clinical pyramidal involvement.31 Changes in cortical excitability (RMT), central cholinergic activity (SAI), or intracortical inhibition/facilitation were not observed in FTD.43,44 Pierantozzi et al.45 reported that early-onset AD patients showed significant low SICI (ISI = 2-3 ms), whereas FTD patients did not.
Based on the above observations, some studies have investigated using TMS to differentiate AD patients from healthy controls and those with other types of dementia. One study used TMS-based assessment of GABAergic and cholinergic neurotransmission to distinguish patients with AD from FTD patients and healthy controls.42 AD showed significant impairment of SAI, and FTD showed dysfunction of SICI-ICF. These findings had high sensitivity and specificity in differentiating patients with each disease from healthy controls. Another multicenter study found that AD and DLB patients showed low SAI, representation of cholinergic deficits, and that FTD and DLB had an abnormality of SICI-ICF associated with GABAergic and glutamatergic circuits.46 Moreover, LICI was most impaired in FTD. These results are related to the biological pathology of neurotransmitters in each neurodegenerative disease, and demonstrate the ability to use TMS parameters to differentiate the diseases with high sensitivity and specificity (Table 1).
It has been suggested that the SAI measure could be used as a tool to identify MCI patients with an increased risk of converting to dementia.47 In studies of MCI patients and healthy controls, SAI was reduced in multiple-domains amnestic MCI, which has the highest risk of converting to dementia, while it did not differ in single-domain amnestic and non-amnestic MCI patients compared with normal cognition.47,48
One study investigated the benefit of acetylcholinesterase inhibitor (AChEi) treatment in changing plasticity within cortical glutamatergic circuits.49 MEP amplitudes were measured during 5-Hz rTMS (which reflects short-term synaptic plasticity) before and after AChEi administration. The application of AChEi for 2 months did not influence the alteration in cortical excitability or short-term synaptic plasticity. Other studies have estimated SAI, which represents central cholinergic activity, and found that the low SAI in AD could be restored by the administration of a single dose of AChEi.37,50 Furthermore, AD patients who showed a large increase in SAI after a single dose of AChEi respond well to long-term treatment.
It appears conclusive that the clinical benefit of AChEi treatment is related more closely to factors including cholinergic activity than to the activity of glutamatergic NMDA receptors. Therefore, evaluating SAI could be useful for predicting the response to AChEi.
Therapeutic interventions
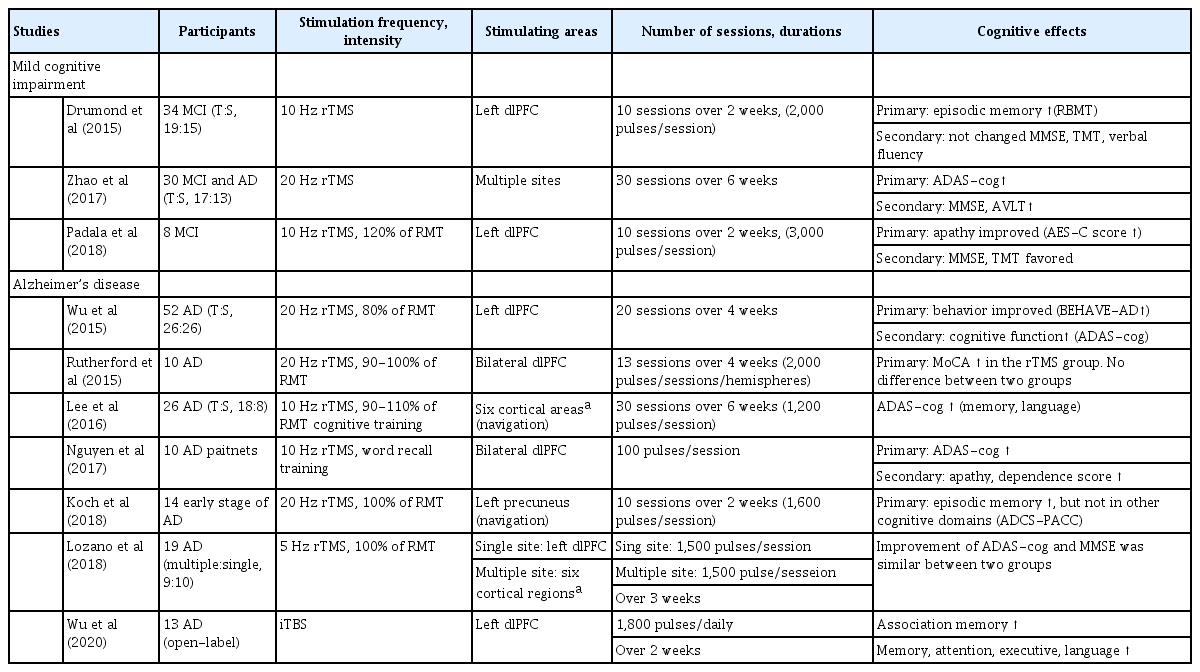
Multiple sessions of rTMS have recently been proposed as a promising treatment for improving cognitive performance in AD (Table 2). rTMS can result in temporal changes in focal cortical excitability producing prolonged changes in neuron integrity. LTP-like cortical plasticity is disrupted in the early stage of AD, whereas high-frequency rTMS can enhance LTP-like cortical plasticity at both the local and network levels.51 Multiple sessions of rTMS induced synaptic plasticity and especially LTP, which was the most important mechanism in learning and memory. rTMS was helpful in restoring brain functions and could reflect the potential of rTMS to recruit compensatory networks that underlie memory-encoding and other cognitive processes.52
Summary of recent rTMS studies of cognitive effects in Alzheimer’s disease and mild cognitive impairment
High-frequency rTMS over the dorsolateral prefrontal cortex (dlPFC) can improve global cognition, action naming, object naming, episodic memory, and information processing speed in patients with AD, MCI, and cognitively normal controls.53-55 Most rTMS studies have focused on the dlPFC, and this area is affected in both the early and late stages of dementia. Researchers have recently applied rTMS to the posterior parietal cortex and precuneus, which were affected in the early stage and had prominent neuropathological abnormalities. The effects of rTMS in various types of dementia are summarized in Fig. 1.
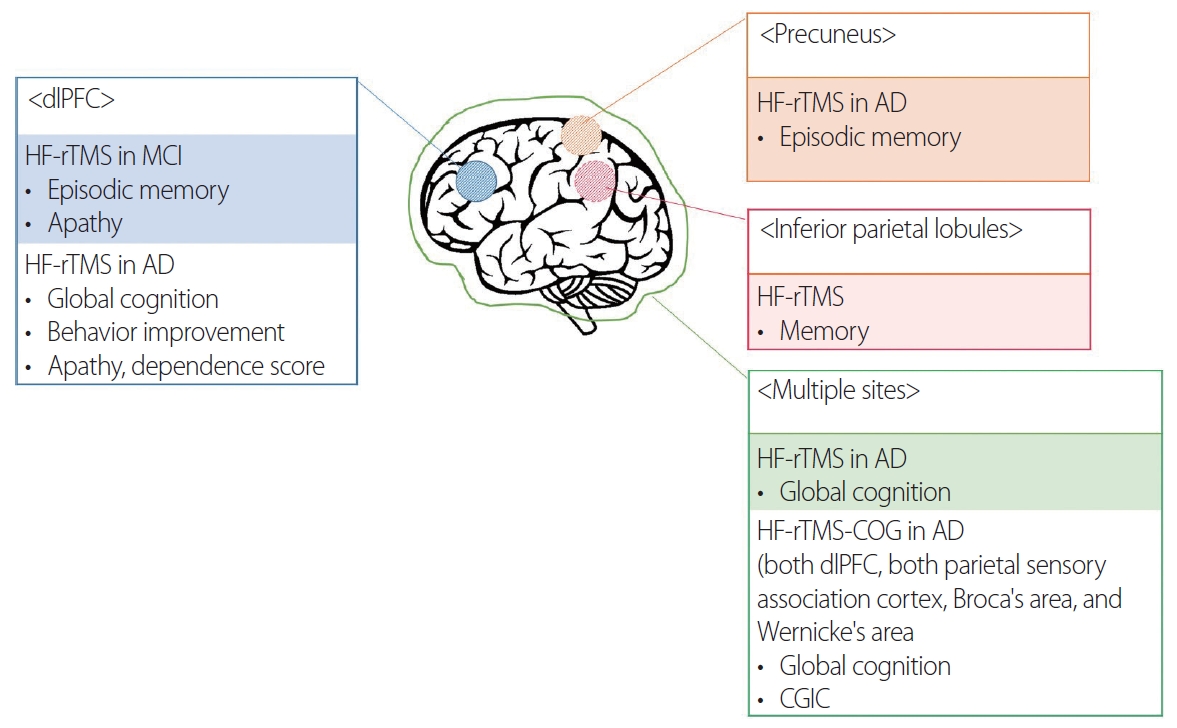
Effects of high-frequency repetitive transcranial magnetic stimulation (HF-rTMS) in Alzheimer’s disease (AD) according to cortical areas. dlPFC, dorsolateral prefrontal cortex; MCI, mild cognitive impairment; CGIC, clinical global impressions of change.
We have reported on the effect of rTMS with cognitive training (rTMS-CT) in patients with mild-to-moderate AD.56 The participants in that study received rTMS in 6 cortical areas and showed cognitive improvement after 30 sessions. The effect was maintained at 6 weeks after the end of the treatment, and was more prominent in the mild-AD group. Similar to our study, other studies have found better cognitive effects of rTMS in the early stage of AD.57 These results might be due to the degree of gray-matter atrophy in AD-related brain regions contributing to the variability of rTMS-induced cognitive aftereffects.58 Patients with AD who received rTMS-CT showed slower cognitive decline than did those who only took AChEis over 3 years.59 That study revealed for how long rTMS-CT could affect the disease course.
rTMS can improve neuropsychiatric symptoms in AD as well as enhance cognitive function. A meta-analysis of the efficacy of rTMS on the neuropsychiatric symptoms of AD found that rTMS could persistently improve the neuropsychiatric symptoms at an early stage after treatment.60 However, following the evidence-based guidelines on the therapeutic use of rTMS,61 further evidence is needed for the use of rTMS as a therapeutic tool in AD and MCI.
TBS is emerging as a more efficient treatment paradigm in terms of time and energy than rTMS.62 TBS involves applying a 50-Hz triplet pulse burst with a 200-ms interburst interval, typically at 80% of the active MT. Continuous TBS (cTBS) delivers 600 pulses for 40 seconds without interruption, while intermittent TBS (iTBS) involves 2-s-long TBS trains repeated every 10 seconds for a total of 20 cycles. cTBS generally decreases cortical excitability for up to 1 hour, while iTBS increases cortical excitability more rapidly than conventional rTMS.24 Several studies have applied iTBS for treating dementia. In an open-label study, 13 patients with AD received 14 sessions of iTBS daily over a 2-week period.63 After treatment, they showed cognitive improvement in memory, attention executive, and language functions, and their behavioral and psychiatric symptoms were also ameliorated.
CONCLUSION
Many studies have investigated TMS over the last 20 years, and many developments have been made in various fields. TMS paradigms can address many neurophysiological states of the neural integrity and functional changes in neurotransmitters in vivo. It is possible that TMS parameters such as the MT, MEP, SICI-ICF, and SAI could be useful biomarkers for diagnosing and differentiating different types of dementia. Moreover, rTMS and TBS can be adjunctive management methods for cognitive decline and abnormal behavior in AD. Since TMS is a painless, and noninvasive tool, future researches could lead to the development of systematic TMS evaluation and treatment protocols for aiding the diagnosis and management of dementia.
Notes
Conflicts of Interest
The authors declare no conflicts of interest relevant to this article.